FAQs
Frequently Asked Questions (FAQ) following the publication in eClinicalMedicine
When we first saw Long COVID patients in our pain clinic, it was initially in the context of treating severe neuropathic pain. These patients had long-term symptoms after COVID-19 and there were hardly any effective treatment options. We therefore treated them with intravenous lidocaine, as is usual for therapy-resistant neuropathic pain. What struck us was that not only the pain, but also other Long COVID symptoms began to decrease: symptoms such as brain fog, post-exertional malaise, sensory overload and autonomic symptoms also clearly decreased.
That pattern, improvement of pain and neurological/cognitive symptoms under one treatment, fit the hypothesis that Long COVID is to an important extent a (neuro-)inflammatory condition, in which inflammatory processes in the nervous system and immune system play a central role. After all, lidocaine is not only a local anesthetic, but also has systemic anti-inflammatory properties, particularly on neuroinflammation. From that clinical picture arose the idea to use lidocaine for a longer period and in a more targeted way.
However, intravenous administration requires hospitalization and continuous monitoring of vital functions, and is therefore not suitable as home treatment. Therefore, the choice was made to develop a subcutaneous formulation of lidocaine with hydroxypropyl-β-cyclodextrin (HP-β-CD), intended to remain present in the lymphatic system longer, provide lower plasma levels and thus reduce the risk of toxicity.
The patients described in the article are not a pre-randomized research population, but seriously ill people who were referred to us by their general practitioner for (pain) treatment. Given the severity and duration of their symptoms, we found it ethically difficult to justify first placing this group in a classic placebo-controlled trial for years. In practice, this means at least one to two years before a first patient may be treated. During that period, these patients would then have no access to a treatment that we already saw clearly improved them clinically.
Instead, we chose a care pathway with very intensive monitoring, where we systematically collected data from the moment of intake. Patients received a wearable (Oura ring) that recorded heart rate, activity and sleep, among other things, and an app in which they reported the severity of their symptoms daily and completed questionnaires every two weeks. Because the production of the lidocaine-HP-β-CD formula had to be started up via a hospital pharmacy, an observation period of an average of four weeks arose before the first injection. During that period, patients did not yet receive medication, but were already being monitored.
Thus, each patient became their own control: we could compare the course during the pre-treatment phase with the course during the treatment phase. Retrospectively, these data were analyzed together with researchers from VU and Amsterdam UMC in an Interrupted Time Series setup. This is an observational design that, if properly executed and in a population with chronically ill patients like this, can approach the statistical power of an RCT, without withholding a potentially effective treatment from seriously ill people for years.
It is important to emphasize that this is the first scientific article about this treatment, which of course does not yet answer all questions. The results show that a significant proportion of patients with severe Long COVID symptoms experience clear improvement in quality of life and symptom burden. These effects range from modest but clearly noticeable progress for the patient, to almost complete disappearance of symptoms. With good injection instructions, patients can moreover administer the subcutaneous injections themselves at home. At the same time, this is an observational study, without a randomized placebo group, conducted in a specialized clinical setting. Methodologically, an interrupted time series design was used with intensive measurements before and during treatment. This design approaches, according to Prof. J.W.R. Twisk (Professor of Statistics and Methodology AUMC), if properly executed in a stable chronic population like this, the statistical power of a randomized trial, but it formally remains an observational study. This means the findings are promising, but still need to be confirmed in an RCT. For formal registration of the treatment, consideration is also being given to the design of such an RCT, in which part of the patients receive active treatment and part receive placebo. Such research can easily take 2 years. Additionally, there are ethical and practical objections to giving placebo to seriously ill patients.
Based on current data, we believe the therapy is sufficiently substantiated to continue treating, especially in specialized centers with experience and digital monitoring. In combination with the large, unanswered care need of seriously ill Long COVID patients, we do not consider it responsible to now put this treatment "on hold" awaiting even more evidence, given the very urgent need for help from thousands of Long COVID patients. This does not mean that additional research is not still needed, on the contrary: randomized studies, longer-term follow-up and better insight into which patients benefit most are essential for further refinement and any formal registration and package assessment. But we see the current results as sufficiently strong to keep the treatment available now for patients for whom other options are lacking, parallel to setting up follow-up research.
It is true that generic lidocaine as a raw material is not expensive. However, the treatment we are publishing about does not involve standard lidocaine, but a specially developed formulation for daily home administration and targeted uptake via the lymphatic system and lymph nodes.
For this application, lidocaine has been:
- Pharmaceutically modified to be preferentially absorbed via the lymphatic system rather than the bloodstream after subcutaneous administration.
- Formulated with specific excipients, in a precisely controlled composition, so that effective immune modulation is possible at low and safe plasma concentrations.
- Made suitable for safe self-administration at home, thereby preventing burdensome and costly hospital treatments.
The medication:
- Is currently prepared on a relatively small scale by a hospital pharmacy,
- This is done according to strict quality and safety standards (GMP/GDP),
- Involves high associated costs for pharmaceutical validation, quality control, packaging, distribution and medical monitoring (partly digital).
It is this small-scale preparation by a hospital pharmacy that explains a significant part of the current costs.
At the same time, preparations are being made to scale up production in the short term in collaboration with an international large-scale compounding facility. Through this scale-up:
- Production processes can be standardized,
- Efficiency increases significantly, and
- As a result, the cost per patient is expected to drop substantially.
In summary: This is not about "expensive lidocaine," but about a formulation suitable for daily home administration that is currently prepared on a small scale and that can become significantly cheaper with international scale-up.
No. This concerns a compounded (magistral) preparation: a common practice where, in collaboration between the treating physician and pharmacist, an existing registered active substance (lidocaine) is prepared with an excipient for a specific route of administration. The active molecule (API) is unchanged. This therefore does not involve a new molecule or a new active substance.
The pharmacy-compounded lidocaine with HP-β-cyclodextrine is not (yet) covered by health insurers, which leads patients and doctors to look for cheaper alternatives.
However, standard lidocaine infusion solution is not suitable for this purpose. In pain medicine, this form of lidocaine is administered exclusively in a hospital setting, under continuous medical supervision. Administration via infusion or injection (intravenous or subcutaneous) can lead to excessively high lidocaine blood levels, carrying a risk of serious side effects. For this reason, this treatment only takes place in a clinical setting.
For the vast majority of long COVID patients, having to travel to a hospital repeatedly is simply too burdensome. To enable safe home treatment, a modified formulation was therefore developed in which the excipient hydroxypropyl-β-cyclodextrine (HP-β-CD) has been added to lidocaine.
This formulation is designed with the purpose of, after subcutaneous administration, lidocaine is preferentially absorbed through the lymphatic system rather than directly into the bloodstream. This enables patients to safely self-inject at home. In addition, HP-β-CD makes the injections less painful and allows the effective dose to be delivered in a smaller injection volume, further reducing the risk of systemic absorption.
The safety, side effects, and outcomes of this modified lidocaine treatment were studied in 103 long COVID patients who were intensively monitored on a daily basis. These findings have recently been published in the scientific literature.
By contrast, home injection of standard lidocaine infusion solution has not been studied and is strongly discouraged on safety grounds. GPs and medical specialists are therefore advised to use lidocaine infusion solution exclusively within a hospital setting.
There is indeed follow-up after stopping or tapering treatment. The published results run until April 2025; since then we continue to follow these patients. We are currently preparing a follow-up publication with data up to 66 weeks. Preliminary analyses show that the positive effects on both physical and mental health remain stable in many cases or even continue to increase, and that a significant proportion of patients have been able to reduce or completely stop the medication without relapse to the old symptom level.
At the same time, precisely because of the impact of these findings, we consider it important to confirm and refine the results in a formal, placebo-controlled Randomized Controlled Trial. Therefore, a RCT is now being prepared.
This is an important misconception. Many people recover in the first months after a COVID-19 infection, but a significant proportion have long-term symptoms. Large studies show that approximately 10-20% of people still have clear symptoms after 12 months that affect daily functioning. When symptoms persist for longer than a year, we see that spontaneous recovery occurs less frequently. For this group, Long COVID is not a brief aftermath, but in many cases a serious, disabling, chronic condition. Our patients had been seriously ill for an average of 2.5 years without signs of improvement with or without therapy. Another misconception is that Long COVID mainly occurs in people who were very seriously ill during the corona crisis and/or were in intensive care. The vast majority of severe Long COVID patients we have treated developed the disease several weeks after a 'regular' COVID-19 infection from which they thought they had almost recovered.
The study in eClinicalMedicine is not an ordinary observational study, but an "interrupted time series" (ITS) study, a method specifically used as an alternative when an RCT is not (yet) feasible. In this design, patients essentially serve as their own control group.
Concretely, this means that for 103 post-COVID patients, a pre-treatment period of an average of 4 weeks was first measured, during which they completed their symptom scores daily and the SF-12 quality of life questionnaire every two weeks. They then continued these same measurements during 36 weeks of treatment with subcutaneous lidocaine-HP-β-CD. This created a highly detailed timeline of symptoms before and during treatment, to which analyses (linear mixed models) were applied.
According to Prof. Jos Twisk, expert in longitudinal data analysis and co-author of the study, the statistical power of evidence of this ITS design approaches that of an RCT, because there are so many repeated measurements and each patient serves as their own control.
In addition to the questionnaires administered via the app, a Global Perceived Effect questionnaire (GPE) was also administered. Patients completed this GPE questionnaire at our clinic during follow-up visits. These took place every seven weeks.
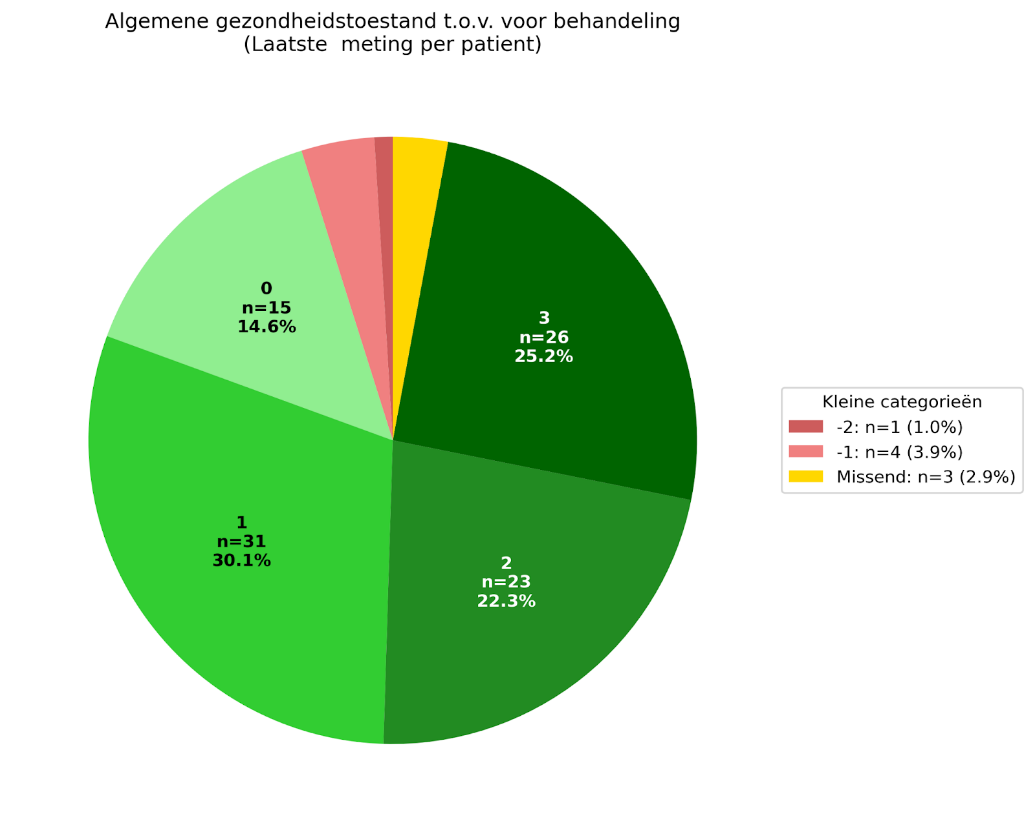
The percentage of 78% is based on the positive scores (1. Somewhat better, 2. Better and 3. Much better) on the question "How do you assess your general health condition compared to before you started treatment?" This percentage is calculated over the complete study group of 103 patients at the 36-week measurement. This approach aligns with the chosen study design, where the complete group of patients over the entire measurement period is always included in all analyses. This includes the last completed GPE scores of all patients, including those who stopped early. There were 30 patients who stopped early. This does not necessarily mean that these patients did not benefit from the treatment. The reasons for stopping early were varied. Some of those who stopped early had given a positive GPE score. In total, 80 of the 103 patients had a positive GPE score. 20 patients fell into the category with negative or neutral GPE score, and GPE data were missing from 3 patients. The positive GPE score, 80 of the 103 patients, corresponds to 78% of the total study group included in the article.
This study used an interrupted time series design. This means that the scores of the same group of patients were measured at multiple time points: before the start of treatment and during treatment. By comparing these measurements over time, you can see whether a change occurs that correlates with the moment when the intervention is started. All patients are included in this analysis, so the found effect reflects an average of the entire group, including dropouts. Within that group, there are of course people who did clearly better, but also people for whom the improvement was smaller or absent. There were also some patients who reported deterioration. For the outcome measures, among others, the Mental Component Summary (MCS) and Physical Component Summary (PCS) scores of a validated questionnaire (the SF-12) were used. The MCS measures mental health, the PCS measures physical health. In the general population, these scores average around 50 points, with a standard deviation of approximately 10 points. Higher scores indicate better health. The smallest change in score noticeable to patients is also called the Minimal Clinically Important Difference (MCID). For Long COVID, there is currently no established MCID. For other chronic conditions, the MCID for the MCS is usually in the order of 3-5 points. This gives an indication of what can be considered clinically relevant, although this still needs to be specifically established for Long COVID. The found improvement of 4.13 on the PCS score and 7 points on the MCS score at 36 weeks is larger than what is usually considered clinically relevant, which indicates that the treatment has provided meaningful improvement for many patients.
If you only look at the numbers +7 (MCS) and +4 (PCS), it doesn't seem spectacular. But those numbers are a summary of the trajectory of all 103 patients combined. Within that group are people who clearly improve, people who show limited improvement, and people who respond little or not at all. We use an intention-to-treat analysis in combination with a linear mixed model. Simply put: the model uses all available measurements from all patients over time and draws an average line through them. Patients who stop early or miss a measurement still continue to count. This is fairer and more realistic than only including people who complete the treatment fully, but it does mean that the group average is lower than the improvement shown by a portion of the patients.
The average of +7 (MCS) and +4 (PCS) therefore does not mean that everyone improves only slightly. In the distribution you can see that a substantial group of patients makes large strides in mental and/or physical health, some improve moderately, and a smaller group changes little to not at all. It is precisely that larger group that clearly improves which is an important reason to continue the treatment.
Additionally, we look at what in medicine is called a clinically relevant change (MCID). For Long COVID, no official threshold has yet been established, but for other chronic conditions, an improvement of approximately 3-5 points on such scales is already considered noticeable and relevant. The improvements we see (+7 and +4 on average across the entire group) are within or above that range. This also aligns with what patients themselves report: 78% report a clear improvement in quality of life.
Given that this involves a chronic disease with long-term severe symptoms, these are meaningful results. Therefore, we conclude that a large proportion of patients show a clear and relevant treatment effect, and this forms a strong reason to continue both the treatment and further research.
For answering this question, the distinction between incidents/events and adverse events is important.
An incident is an undesirable event during treatment or in the care process (a symptom that occurs, an injection that unintentionally ends up in a blood vessel, a bruise after injection, etc.). Only later is it assessed whether an incident is an adverse event: Adverse Event (AE): adverse event regardless of whether there is a link to the medication, an Adverse Drug Reaction (ADR): adverse event probably or confirmed to be caused by the medication, or a Serious Adverse Event (SAE)/Serious Adverse Drug Reaction (SADR), a serious adverse event with serious consequences, such as death, a life-threatening situation, (prolonged) hospitalization or permanent damage (ICH guidelines for Good Clinical Practice).
In total, 64,281 injections were prescribed in the period up to 36 weeks after the start of treatment. During this period, 73 incident reports were received of (partial) intravascular injection (into the bloodstream), from 34 patients in total. An injection into the bloodstream can lead to symptoms such as: dizziness, tingling around the lips, tinnitus, headache, and (tendency to) fainting. These symptoms were short-lived and reversible. Some patients subsequently indicated experiencing PEM symptoms comparable to excessive exertion. Four incidents led to a 112 emergency call, one of which resulted in an emergency room visit by ambulance. After evaluation by an emergency room physician, the patient could return home. Based on the aforementioned ICH guidelines, none of these incidents were classified as an SAE. The incident where the patient was assessed in the emergency room was reported to the Health and Youth Care Inspectorate (IGJ). After adjusting the injection instructions (including injecting over 120 seconds and rotating injection sites), the number of reported intravascular injections decreased by 83%.
The skin reactions that can occur with daily subcutaneous injections are classified in the article as "mild adverse events" because they do not cause permanent damage. In clinical research, adverse events are categorized by severity: mild, moderate, or severe (severity grading). Skin reactions such as redness, swelling, bruising or hardening fall into the mild adverse events category. This does not mean that these symptoms cannot be bothersome for patients. However, many patients found the skin reactions from the daily injections not troublesome in proportion to the experienced improvement of their symptoms. The skin problems usually disappear within several weeks to months after tapering/stopping the injections. Skin reactions are to be expected with frequent subcutaneous injections, as is also known from insulin or anticoagulation treatments, for example. However, these reactions do require attention. Over time, various strategies have been implemented to support the skin as well as possible and limit problems, such as cooling, massage, lymphatic drainage, the use of grid plasters, and regularly changing injection sites.
In the study we describe the results up to and including 36 weeks after the start of treatment, but in practice the treatment duration varies per patient. Patients are usually treated for several months; treatment continues until someone has stably recovered to approximately 80% of functioning before the onset of the disease. At that point, in consultation between patient and treating physician, tapering is started and, where possible, stopped. The treatment consists of subcutaneous injections with lidocaine-HP-β-CD, which patients administer themselves at home according to a fixed schedule. During treatment they are intensively monitored: patients report the severity of their symptoms daily via a web application, complete more extensive questionnaires at fixed moments, and come for a clinical check approximately every six weeks. This way, the course of symptoms, the moment of tapering and the safety of treatment can be closely monitored.
For Long COVID, it is estimated that worldwide approximately 450 million people suffer from chronic, non-spontaneously improving symptoms. In the Netherlands, it is estimated to involve 350,000-400,000 people (approximately 2-2.5% of the population) with long-term symptoms, of whom around 100,000 are seriously ill.
Many of these seriously ill patients (partially) disappear from view, because no reliable diagnostic test is yet available. Medical specialists such as neurologists, cardiologists and pulmonologists usually find few abnormalities in blood tests or imaging that can explain the severity of symptoms. As a result, the condition has long been approached either as a difficult-to-interpret multisystem disease with various possible underlying mechanisms, or, incorrectly, mainly interpreted as psychosomatic, with major medical, social and financial consequences for patients.
The growing insight that Long COVID probably has an important immunological component and appears treatable in at least part of patients, is expected to usher in a new phase, both for patients and for healthcare providers. The therapy described here is specifically aimed at people with severe, disabling symptoms; that is a very large group, in the Netherlands and worldwide, for whom hardly any effective treatment options are currently available.
Excellent Care Clinics (ECC) has been working for four to five years to improve a treatment for Long COVID step by step. During this period, much time, energy and money have been invested, without support from the government (subsidy) or a large pharmaceutical company. Based on this development, the decision was made to patent the findings. This does not concern lidocaine itself, which is an old and inexpensive drug whose patents have long expired, but the new composition and administration method specifically targeted at the lymph nodes/immune system.
The purpose of these patents is to transfer them to an international pharmaceutical company once the new mechanism is sufficiently demonstrated with good scientific research. Such a party can bear the enormous costs necessary for international registration, then produce it on a large scale at low cost per patient, and thereby ensure that the treatment ultimately becomes available to as many people as possible, also outside the Netherlands. Without patent protection, no party will take on the development and registration costs. ECC is a medical enterprise that, according to principles of good governance, has the duty to keep the healthcare business economically healthy. This is necessary to guarantee continuity of care, pay healthcare personnel and continue to invest in new equipment and further innovation. In that light, it is desirable that the patents also contribute to the invested money being earned back over time. This is not about profit maximization, but about a condition to further develop treatments, make them affordable and available on a large scale, and guarantee continuity of good care.
In research, you can measure two types of outcomes: objective measurements and subjective measurements. Objective measurements are, for example, blood tests, scans, or other biomarkers that are independent of how someone feels. Subjective measurements are questionnaires in which patients themselves indicate how their symptoms, functioning, and quality of life are progressing.
For Long COVID, there are currently no reliable objective measurements available. There is not yet a blood test, scan, or other biomarker that can clearly show whether someone has Long COVID and whether a treatment works. This is being researched worldwide, but these measurements are not yet sufficiently developed or validated.
Long COVID is defined by international experts as a chronic symptom disorder following SARS-CoV-2 infection, with a wide range of symptoms (fatigue, cognitive problems, exercise intolerance, pain, etc.) that strongly determine what someone can still do in daily life. Those symptoms are by definition what the patient feels and experiences. Therefore, questionnaires about: physical functioning (PCS, physical subscale SF-12), mental functioning (MCS, mental subscale SF-12), and global improvement (GPE / "how are you doing now compared to before?"), are currently the most direct and clinically relevant way to measure whether someone is actually improving.
This is why subjective outcomes are now the standard in Long COVID research. Major international studies work with these, and the European Medicines Agency (EMA) also indicates that these outcomes are currently an acceptable and appropriate way to measure treatment effects in Long COVID.
We align with what is internationally considered the best available method: symptom scores and quality of life questionnaires as primary outcomes. These are also exactly what is used in recent major Long COVID studies. In follow-up research (such as the planned RCT), we do want to measure biomarkers alongside these subjective outcomes, so that hopefully in the future we can also use more objective outcome measures.
In short: because no objective measurements for Long COVID yet exist, questionnaires and patient-reported outcomes are currently the worldwide standard and the best available method to determine whether someone is actually improving.
No. This involved regular care: patients were treated according to standard clinical practice within our clinic. Only after the initial treatment results became available were these existing data retrospectively analyzed by researchers from Vrije Universiteit and Amsterdam UMC. Because there were no additional procedures, no randomization, and no research intervention, this fell under observational real-world research and no WMO declaration was required.
WMO (Medical Research Involving Human Subjects Act): Dutch law that determines when prior review by a medical ethics committee is mandatory for research in which subjects receive additional procedures, measurements, or behavioral rules that do not belong to regular treatment.